Search results
Search for "sacrificial electron donor" in Full Text gives 15 result(s) in Beilstein Journal of Organic Chemistry.
Selectivity control towards CO versus H2 for photo-driven CO2 reduction with a novel Co(II) catalyst
Beilstein J. Org. Chem. 2023, 19, 1766–1775, doi:10.3762/bjoc.19.129

- purpose, three main components are needed: a photosensitizer (PS), which acts like a light-antennae harvesting system in natural photosynthesis, a catalyst (Cat.), reacting directly with CO2 after being reduced, and a sacrificial electron donor (SeD). When the involved (photo)catalysts are homogeneous
- developing the major components of a photocatalytic system for CO2 reduction, such as the photosensitizer (PS), the catalyst, and the sacrificial electron donor (SeD). Nevertheless, the solvent and eventual additives play an important role too [6], as they can influence the (photo)redox properties of the
- [20][21][41]. In addition, the benzimidazolidine derivative, BIH (1,3-dimethyl-2-phenyl-benzo[d]imidazolidine) (shown in Figure 1) suited well as a sacrificial electron donor, because of its high reducing power [47]. The photocatalytic experiments were performed under 420 nm light irradiation unless
Enabling artificial photosynthesis systems with molecular recycling: A review of photo- and electrochemical methods for regenerating organic sacrificial electron donors
Beilstein J. Org. Chem. 2023, 19, 1198–1215, doi:10.3762/bjoc.19.88
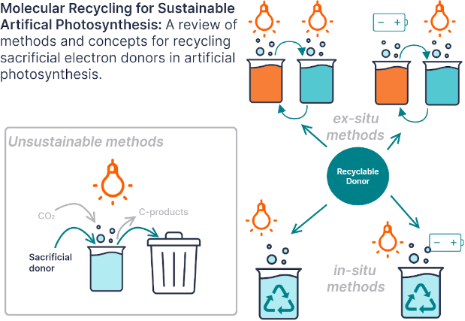
- Grace A. Lowe van ’t Hoff Institute for Molecular Sciences (HIMS), Universiteit van Amsterdam (UvA), Science Park 904, Amsterdam, 1098 XH, The Netherlands 10.3762/bjoc.19.88 Abstract This review surveys advances in the literature that impact organic sacrificial electron donor recycling in
- sacrificial electron donors, and for researchers interested in designing new redox mediator and recyclable electron donor species. Keywords: artificial photosynthesis; photocatalysis; redox couple; sacrificial electron donor; solar fuels; Introduction Artificial photosynthesis research has resulted in the
- material that shuttles electrons from one species to another through a series of chemically reversible reduction and oxidation reactions. In contrast, a sacrificial electron donor is a species that is oxidized to reduce another species and is consumed rather than regenerated. If a redox mediator is not re
Photoredox catalysis harvesting multiple photon or electrochemical energies
Beilstein J. Org. Chem. 2023, 19, 1055–1145, doi:10.3762/bjoc.19.81
- of iron CT states (in the nanosecond domain) enabled by the relatively longer lifetimes of e.g. Fe–NHC complexes [97][98][99][100]. In particular, the Wärnmark group reported two sets of conditions with and without Et3N as a sacrificial electron donor, to achieve reductive and oxidative quenching
- lower catalyst loading. The requirement for sub-stoichiometric amounts of the sacrificial electron donor can be justified. While the work of Wärnmark is remarkable on a conceptual level, the protocol is still limited by the reductive power of the excited state and thus restricted to rather activated
- reduction to PC•− ensures higher concentrations that are directly user-influenced. Upon activation, PC1 could successfully reduce various aryl halides generating borylated products in modest to excellent (30–99%) yields. Control experiments confirmed that light, catalyst and DBU as a sacrificial electron
Insight into functionalized-macrocycles-guided supramolecular photocatalysis
Beilstein J. Org. Chem. 2021, 17, 139–155, doi:10.3762/bjoc.17.15

- ], the [Co(dmgH)2(4-ppy)2]NO3 (27, dmgH2 = dimethylglyoxime, 4-ppy = 4-phenylpyridine) guest, and the EY photosensitizer, respectively (Figure 16) [49]. When mixed with the sacrificial electron donor TEOA in an anaerobic H2O/CH3CN 1:1 solution, this supramolecular system 27@CB[7]/EY could realize an
Heterogeneous photocatalysis in flow chemical reactors
Beilstein J. Org. Chem. 2020, 16, 1495–1549, doi:10.3762/bjoc.16.125

- interest in g-C3N4 photocatalysts was generated in 2009 by Wang, Domen, and co-workers, who reported the metal-free photolysis of water was possible with the all-organic semiconductor material, a sacrificial electron donor, and visible light irradiation [120]. Prior to this report, poly(p-phenylene) had
- photocatalyst for the reduction of aryl halides [193]. The material could undergo consecutive photoinduced electron transfers (ConPET) in which the material enters an excited state and is reduced by a sacrificial electron donor (NEt3). The resulting Zn-PDI radical anion then undergoes a second photon absorption
Photocatalysis with organic dyes: facile access to reactive intermediates for synthesis
Beilstein J. Org. Chem. 2020, 16, 1163–1187, doi:10.3762/bjoc.16.103
- each of these steps, the role of A or D is assumed by a redox-active agent, either the substrate, a sacrificial electron donor/acceptor, or a reactive intermediate. This approach, usually named photoredox catalysis, has known a remarkable growth in the last decade and has given access to both neutral
- ), in the presence of the sacrificial electron donor DIPEA, can reduce these species under green light irradiation. The ensuing decarboxylation provides a C(sp3) radical, which undergoes a radical conjugate addition with a suitable Michael acceptor 4.2, providing the desired alkylation products 4.3. A
- combination of eosin Y (OD13) with a sacrificial electron donor can trigger the reductive debromination of several α-carbonyl halides [53]. Riboflavin (OD11) [54] and thiaporphyrin [55] have been applied as well as organic photocatalysts for similar reductive dehalogenations. Hydrogen atom transfer
Visible-light-induced addition of carboxymethanide to styrene from monochloroacetic acid
Beilstein J. Org. Chem. 2020, 16, 398–408, doi:10.3762/bjoc.16.38
- generation of hydrated electrons and applied that for the dehalogenation of monochloroacetic acid [32][33]. However, to the best of our knowledge, a photoredox catalyzed application of monochloroacetic acid without using a sacrificial electron donor is not known. Interestingly, monochloroacetic acid also
- nucleophilic substitution and resulted in very low yields. Sodium ascorbate was also added to investigate whether a sacrificial electron donor could increase the yields, but this was not observed. Interestingly, the reaction with sodium ascorbate favored the formation of lactone 1 over linear acid 3. The exact
Cobalt- and rhodium-catalyzed carboxylation using carbon dioxide as the C1 source
Beilstein J. Org. Chem. 2018, 14, 2435–2460, doi:10.3762/bjoc.14.221

- et al. reported the Rh-catalyzed hydrocarboxylation of alkenes driven by visible-light irradiation conditions in the presence of a photoredox catalyst (Scheme 37) [75]. A model reaction using 4-cyanostyrene (40a) was carried out using iPrNEt2 as a sacrificial electron donor in the presence of [Ru(bpy
Preparative semiconductor photoredox catalysis: An emerging theme in organic synthesis
Beilstein J. Org. Chem. 2015, 11, 1570–1582, doi:10.3762/bjoc.11.173
- transformations an excess of a sacrificial electron donor (reductive quencher) is needed to scavenge the VB holes and prevent e−/h+ recombination. Alcohols and amines have been successfully deployed in this role. Reductions of nitroaromatics took place effectively in photoactivated aqueous TiO2 slurries, with
An integrated photocatalytic/enzymatic system for the reduction of CO2 to methanol in bioglycerol–water
Beilstein J. Org. Chem. 2014, 10, 2556–2565, doi:10.3762/bjoc.10.267

- . Figure 10 shows the influence of the nature of the electron donor and the concentration of the electron mediator, on the reduction rate. The NADH regeneration rate exhibits a strong dependence on the concentration of glycerol, which plays the crucial role of a sacrificial electron donor. Figure 10 also
Visible light photoredox-catalyzed deoxygenation of alcohols
Beilstein J. Org. Chem. 2014, 10, 2157–2165, doi:10.3762/bjoc.10.223
- light in the presence of [Ir(ppy)2(dtb-bpy)](PF6) as visible light photocatalyst and Hünig’s base as sacrificial electron donor in an acetonitrile/water mixture generally gave good to excellent yields of the desired defunctionalized compounds. Functional group tolerance is high but the protocol
- byproducts. Related to this work, Stephenson et al. elegantly succeeded in the direct deoxygenation of alcohols by their in situ conversion to iodides using triphenylphospine and iodine followed by visible light-mediated reduction with amines as stoichiometric sacrificial electron donor and fac-Ir(ppy)3 (ppy
- ; dtb-bpy = 4,4′-di-tert-butyl-2,2′-bipyridine] as photocatalysts, Hantzsch ester (diethyl 1,4-dihydro-2,6-dimethyl-3,5-pyridinedicarboxylate) as hydrogen donor, and iPr2NEt as sacrificial electron donor in DMF (Scheme 3). Light generated from a high power LED was channeled into the reaction solution in
Organic synthesis using photoredox catalysis
Beilstein J. Org. Chem. 2014, 10, 1097–1098, doi:10.3762/bjoc.10.107

- catalysis or light-driven charge separation, which leads to an energy harvesting process by taking advantage of the reduction products and filling the holes by a sacrificial electron donor, water. Fortunately, we can use the waste product from this process, oxygen, for breathing. For applications in organic
The chemistry of amine radical cations produced by visible light photoredox catalysis
Beilstein J. Org. Chem. 2013, 9, 1977–2001, doi:10.3762/bjoc.9.234
- to reductively quench the photoexcited state while they are oxidized to amine radical cations. This single-electron transfer process was investigated intensively in the late 1970s and early 1980s because amines were used as a sacrificial electron donor in water splitting [31][32] and carbon dioxide
- chemistries that have focused on the use of amines as a sacrificial electron donor only or as a hydrogen radical donor only will not be discussed in the review. These chemistries have been recently reviewed [22][23][35][36][37][38][39][40][41][42]. Photooxidation of amines to amine radical cations can also be
Synthesis of rigidified flavin–guanidinium ion conjugates and investigation of their photocatalytic properties
Beilstein J. Org. Chem. 2009, 5, No. 26, doi:10.3762/bjoc.5.26
- the presence of sacrificial electron donor substrates, such as aliphatic amines, flavins can photoreduce nitro arenes to anilines under blue light irradiation (Scheme 4). 4-Nitrophenyl phosphate was used as a substrate for photoreduction in water and in acetonitrile. The results summarized in Table 2
Towards practical biocatalytic Baeyer- Villiger reactions: applying a thermostable enzyme in the gram- scale synthesis of optically- active lactones in a two-liquid- phase system
Beilstein J. Org. Chem. 2005, 1, No. 10, doi:10.1186/1860-5397-1-10
- effective stoichiometric sacrificial electron donor, resulted in significantly decreased stability of both enzymes used. The maximum concentration of isopropanol under which both enzymes show optimal activity was found to be 5% (v/v). In order to enhance conversion, we added surplus reducing equivalents in









































































































































































































































































































































































